Should we be concerned over the involvement of unconscious patients in medical research in New Zealand?
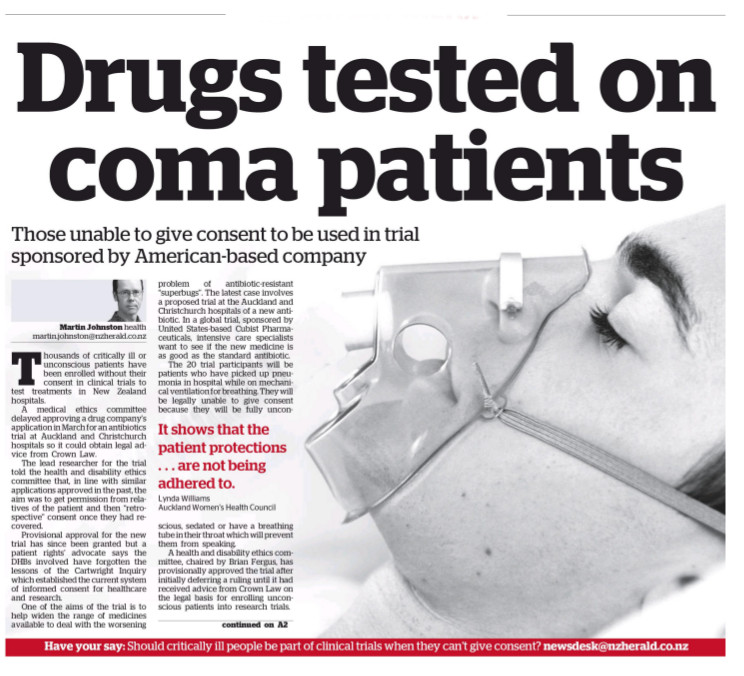 The front page of today’s New Zealand Herald carried a story on a company seeking ethical approval for an antibiotics ‘non-inferiority’ trial involving comatose patients.
The front page of today’s New Zealand Herald carried a story on a company seeking ethical approval for an antibiotics ‘non-inferiority’ trial involving comatose patients.
The Health and Disability Ethics Committee minutes and the Auckland Women’s Health Council newsletter which sparked the story are published on the Herald Online page linked above.
The Science Media Centre contacted experts for further comment on the story. Feel free to use these quotes in your reporting. If you would like to contact a New Zealand expert, please contact the SMC (04 499 5476; smc@sciencemediacentre.co.nz).
Professor Gareth Jones, Bioethics Centre, University of Otago, comments:
“While one can sympathize with the researchers’ predicament when dealing with unconscious patients, enormous care is required to ensure that the best interests of the patients come first. Is the treatment likely to benefit the patient far more than any other available treatment?
“The notion of ‘retrospective consent’ is at odds with that of ‘informed consent’. Retrospective consent is not informed, and it should not be made to appear as though it is. Even if the patient consents after the event, they have been placed in an untenable position, since they are not able to do otherwise.
“Retrospective consent also sets a dangerous precedent, since once normalized, it will legitimize a whole raft of other cases that may bear little resemblance to the current antibiotics research.”
Associate Professor Tim Dare is Head of Department, Philosophy, University of Auckland, and a Barrister and Solicitor of the High Court of New Zealand. He comments:
“I was Chair of one of New Zealand’s busiest Health and Disability Ethics Committees (HDEC) for many years and was until recently Chair of the Health Research Committee Ethics Committee – the committee which has monitors and approves the HDECs.
“I am not aware of the detail of the current study, and it is difficult to give comprehensive advice without those details. However, in my view, the suggestion that research on ICU patients unable to consent can never be justified is incorrect.
“Every international instrument regulating health research recognises that such research might be occasionally be justified:
Declaration of Helsinki:
Para. 26. Research on individuals from whom it is not possible to obtain consent, including proxy or advance consent, should be done only if the physical/mental condition that prevents obtaining informed consent is a necessary characteristic of the research population. The specific reasons for involving research subjects with a condition that renders them unable to give informed consent should be stated in the experimental protocol for consideration and approval of the review committee. The protocol should state that consent to remain in the research should be obtained as soon as possible from the individual or a legally authorized surrogate.
Convention on Human Rights and Biomedicine (add. Protocol art.19):
- The law shall determine whether, and under which protective additional conditions, research in emergency situations may take place when a person is not in a state to give consent and because of the urgency of the situation , it is impossible to obtain in a sufficiently timely manner, authorization from his/her representative or an authority or person or body which would be called upon to give authorisation
- The law shall include the following specific conditions:
- research of comparable effectiveness cannot be carried out on persons in nonemergency situations;
- the research project may only be undertaken if it has been approved specifically for emergency situations by the competent body;
- any relevant previously expressed objections of the person known to the researcher shall be respected;
- acceptance of group benefit if there is minimal risk and burden for the patient
- Persons participating in the ER project or if applicable their representatives shall be provided with all relevant information concerning their participation as soon as possible. Consent or authorization for continued participation shall be requested as soon as reasonably possible
“These cases will almost always be difficult, but the clearest will be where there is genuine uncertainty as to which of two therapeutic options is best. If that is the case, the Code of Patients’ Rights requirement to give priority to the patient’s ‘best interests’ (Right 7(4)(a) will be unhelpful. If there is an important question to be settled, and it cannot be settled using another patient group who could consent, it may be appropriate to conduct approved research using participants who cannot consent.”